What Is The Importance Of Cholesterol Uptake By The Liver For Reverse Cholesterol Transport
Following transport through the plasma compartment, the next step in RCT is delivery of cholesterol from macrophages to the liver. SR-BI is the key receptor responsible for the selective uptake of CEs from HDL into the liver, and hepatic SR-BI has been recognized as a positive regulator of RCT . Consistent with the effects on experimental atherosclerosis, hepatic SR-BI overexpression resulted in more macrophage-derived cholesterol being excreted into the feces , whereas macrophage RCT is clearly impaired in the total absence of SR-BI as well as when SR-BI is exclusively deleted in the liver . Although one study suggested that introduction of CETP can correct the adverse phenotype regarding macrophage RCT in SR-BI knockout mice by shuttling HDL-associated CEs to apoB-containing lipoproteins for receptor-mediated hepatic uptake , this was not confirmed by subsequent research . These differences might be related to the means of CETP overexpression used in these studies, either by AAV or by transgenic overexpression using a construct with the natural flanking regions .
Atherosclerosis Prevention And Regression
A chronic inflammatory disease, atherosclerosis begins with the accumulation of apoB-containing lipoproteins and their cholesterol in the artery wall. In response to arterial lipoprotein/lipid buildup and retention, macrophages are recruited to the intima and take up the modified lipoproteins and their lipids by multiple processes, leading to the formation of foam cells that secrete inflammatory mediators and promote the development of early atherosclerotic lesions. These lesions develop into disease-causing advanced plaques in the process commonly referred to as atherosclerosis progression. Once advanced atherosclerotic plaques are established, the process by which they undergo a reduction in one or more standard parameters is termed atherosclerosis regression. Macrophage RCT is the mechanism by which atherosclerotic plaques may rid themselves of cholesterol, and, as noted earlier, is still considered as an essential target to inhibit atherosclerosis progression and promote atherosclerosis regression.
Reverse Cholesterol Transport: A Cholesterol Removal System
| Font : A-A+ |
A process in which accumulated cholesterol is removed from tissues is called reverse cholesterol transport, including the artery wall and transported back to the liver for excretion. Little is known about how cholesterol is removed from peripheral tissues, but a better understanding of these mechanisms could help in the development of therapies that treat atherosclerosis and other cholesterol-related disorders. In this issue of the Journal of Clinical Investigation, researchers led by Gwendalyn Randolph and colleagues at Washington University in St. Louis examined the role of the lymphatic system in RCT.
Read Also: Canned Tuna Good For Cholesterol
Vascular Smooth Muscle Cells
While much of the focus on the early steps of RCT has been on defining mechanisms of efflux from macrophages, there have also been investigations on vascular smooth muscle cells . VSMC plasticity in atherosclerosis is well recognized e.g., in the media of atherosclerotic arteries, they are considered to be contractile and can become proliferative, migrate to the intima, where they are synthetic, and exhibit the loss of a number of markers of the VSMC in the contractile state, such as smooth muscle cell actin and myosin heavy chain. Though it has been long appreciated that VSMC in the intima can also take up cholesterol through a variety of pathways,,, phenotypic changes in these VSMC-foam cells at the molecular level had not been systematically studied. In 2003, it was shown that loading mouse primary VSMC with cholesterol in vitro resulted in the concurrent loss of VSMC marker expression and the gain of macrophage-associated gene expression.
If the current speculation that transitioned VSMC have negative contributions to atherosclerosis are true, then the value of restoring cholesterol efflux to intimal VSMC becomes clear. An interesting question arises: how do the efflux capacities of distinct foam cell populations differ from one another? The answer would have implications in designing therapeutic strategies to target all or a subset of foam cells in the plaque to maximally promote RCT.
What Gets Stored In A Cookie
This site stores nothing other than an automatically generated session ID in the cookie no other information is captured.
In general, only the information that you provide, or the choices you make while visiting a web site, can be stored in a cookie. For example, the site cannot determine your email name unless you choose to type it. Allowing a website to create a cookie does not give that or any other site access to the rest of your computer, and only the site that created the cookie can read it.
Read Also: How Much Cholesterol In Pork Chops
Enhancing Reverse Cholesterol Transport
Reverse cholesterol transport , a mechanism by which excess cholesterol in peripheral tissues is transported to liver for biliary excretion, slows foam cell formation and development of atherosclerosis . n3 fatty acids beneficially affect high density lipoproteins remodeling through lecithin cholesteryl acyl transferase and cholesteryl ester transfer protein , facilitating scavenger receptor B1 and LDLr mediated hepatic uptake of plaque-derived excess cholesterol . Dietary supplementation of fish oil promoted RCT by enhancement of hepatic excretion of macrophage-derived and HDL-derived cholesterol . Fish oil increased the gene expression of Abcg5/g8, key proteins regulating hepatic cholesterol secretion into bile, and also downregulated intestinal Npc1l1, which reduces intestinal reabsorption of biliary HDL-derived cholesterol . As reviewed previously, pharmacological and genetic modulation of AA metabolome might also affect RCT. n6 PUFA were shown to lower plasma LDL-cholesterol and plasma total cholesterol to HDL-cholesterol ratio .
Raul Cavalcante Maranhão, … Protásio Lemos da Luz, in, 2018
A Mouse Model Of Dysfunctional Hdl Due To High Hdl
Mice deficient in the HDL receptor SR-B1 are robust models of dysfunctional high plasma HDL-C levels. Compared to wild type mice, HDL in SR-B1/ mice is larger and richer in FC plasma HDL levels of SR-B1/ mice are two-times higher than those of WT.29 SR-B1/ mice are characterized by multiple metabolic defects, such as abnormal platelet and erythrocyte structure and function and infertility among female mice.30 Moreover, when put on a high-fat, high-cholesterol diet, the mice develop atherosclerosis despite a doubling of the HDL concentration and without a meaningful change in the plasma concentration of apoB-containing lipoproteins.31 Thus, while it is by far the most abundant plasma lipoprotein among SR-B1/ mice, some property of HDL is toxic. We reiterate our hypothesis that the pathological quality of SR-B1/ HDL is a high HDL-FC bioavailability that is supported by a high plasma HDL concentration . Comparison of the aforementioned mol values with the data of Figure 3 suggests that the HDL of SR-B1/ mice would strongly support FC influx into cells. Notably, the lipid-lowering drug probucol restores some function, including fertility.30,32 However, this is a partial effect in that probucol reduces the atherogenicity of HDL by reducing its plasma concentration without profoundly affecting HDL-FC content.
Recommended Reading: Does Shrimp Have High Cholesterol
What Proteins Influencing Macrophage Cholesterol Metabolism Are Relevant For Reverse Cholesterol Transport
The first important step in the RCT pathway comprises the removal of excessive cholesterol from macrophage foam cells. The rate of cholesterol movement from macrophages to plasma is determined in the first place by the transport capacity of the macrophage. Cholesterol can be effluxed from the macrophage only in the unesterified or free form, but not as cholesteryl ester . CEs stored in cytoplasmic lipid droplets of macrophages are hydrolyzed by a neutral cholesteryl ester hydrolase , and increased CE hydrolysis in lipid-laden macrophages by overexpression of human nCEH resulted in enhanced efflux of cholesterol . However, such a manipulation is also likely to impact the cholesterol loading of the macrophages used in the RCT experiment. Nevertheless, in vivo the movement of radiolabeled cholesterol from macrophages into feces was significantly higher from macrophages expressing human nCEH , suggesting that efficient hydrolysis of intracellular CEs in macrophages is critical for the first step in RCT.
How Can Reverse Cholesterol Transport Be Quantified
Initial attempts to quantify RCT used mass measurements of centripetal cholesterol flow from extrahepatic organs to the liver . In addition, isotope techniques were employed to assess the dilution of an administered tracer over time by tissue-derived cholesterol . However, all of these methods are not able to specifically trace cholesterol derived from macrophage foam cells, a small but highly relevant pool for atherosclerotic cardiovascular disease .
In 2003 the RCT field took up speed after Rader and colleagues introduced a novel in vivo method to specifically trace the movement of cholesterol from macrophages to plasma, liver, and feces . Briefly, macrophages are loaded in vitro with modified low density lipoproteins and 3H-cholesterol to generate macrophage foam cells. After an equilibration period, the macrophages are injected intraperitoneally into recipient mice. Plasma samples are taken on several time points, and feces are collected continuously during the duration of the experiment. Although this only represents a fraction of the total injected dose within the time frame of a given experiment, completed RCT is defined as the amount of 3H-tracer originating from macrophages that is recovered within feces. Of note, a potentially new experimental approach with macrophage loading in vivo using 3H-cholesteryl oleate-labeled oxidized LDL was recently communicated at scientific conferences , which will, however, not be further discussed in the present review.
Figure 2
Read Also: Potatoes And Cholesterol
Transhepatic Reverse Cholesterol Transport
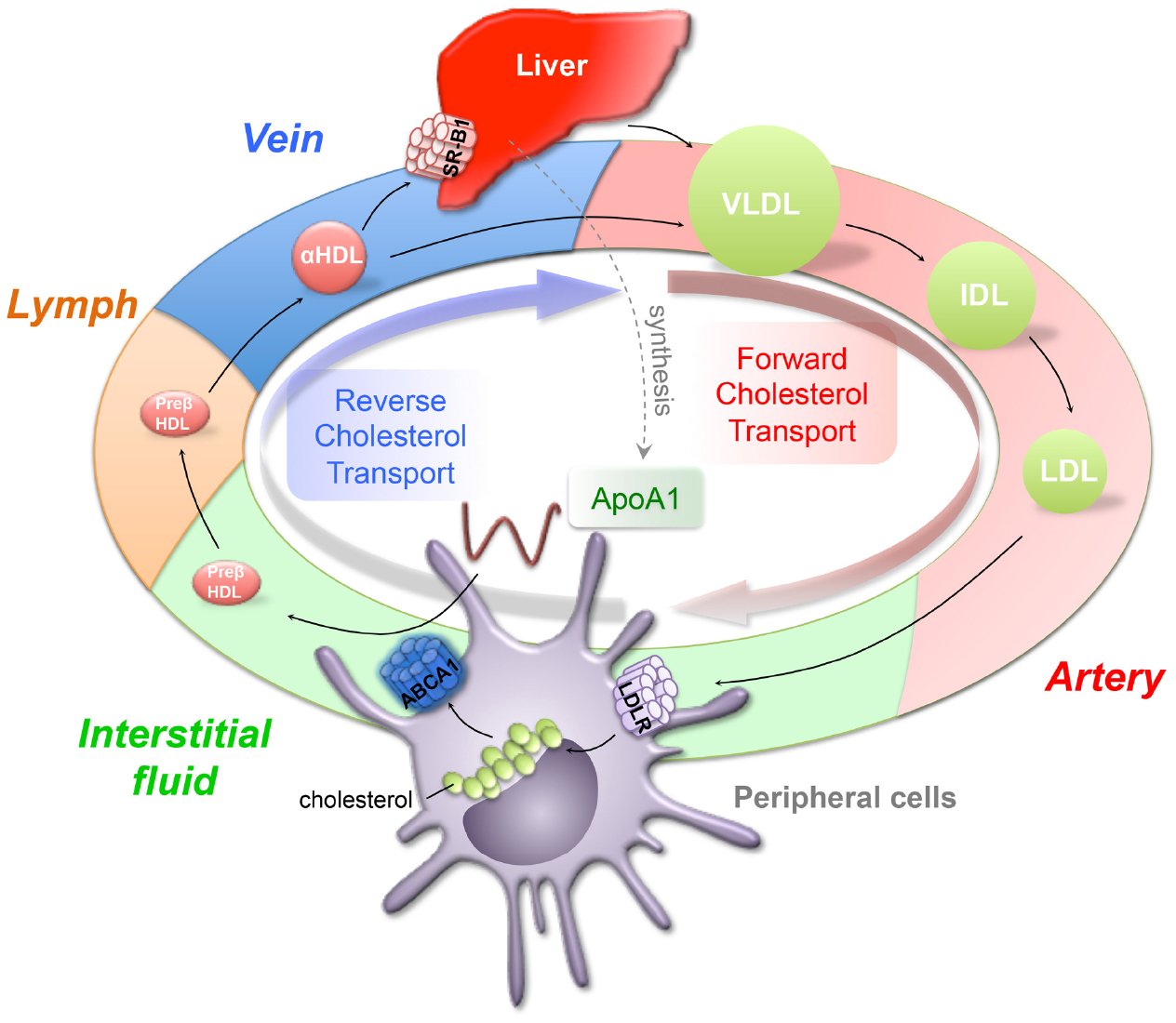
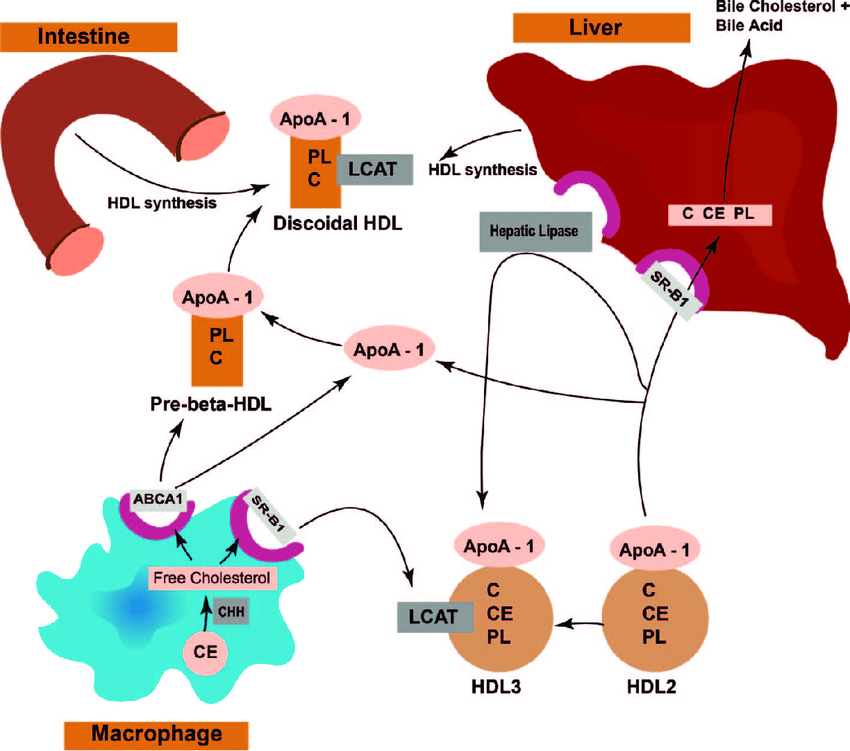
HDL is thought to elicit a cardioprotective effect through its role in reverse cholesterol transport , the transfer of cholesterol from macrophages in the arterial wall to the liver for intestinal disposal . The initial RCT stepcholesterol efflux from macrophages to apoAI via the ATP-binding cassette transporter member 1 forms nascent HDL. This is followed by esterification of HDL free cholesterol by lecithin:cholesterol acyltransferase , which forms mature, spherical HDL with a cholesteryl ester core. Finally, HDL lipids, including FC and CE, are selectively removed by the hepatic HDL receptor, scavenger receptor class B type 1 , leaving a remnant of mostly phospholipid and protein and lipid-free apoAI.21,22
Traditional reverse cholesterol transport model comprises three steps: macrophage free cholesterol efflux to apolipoprotein AI via the ATP-binding cassette transporter A1 yields nascent high-density lipoprotein nHDL esterification by lecithin:cholesterol acyltransferase gives spherical HDL and selective hepatic uptake of HDL cholesterol ester. SR-B1: scavenger receptor class B type 1
Reverse Cholesterol Transport Pathway And Cholesterol Efflux In Diabetic Retinopathy
Xinyuan Zhang
1Beijing Institute of Ophthalmology, Department of Ophthalmology, Beijing Tongren Hospital, Capital Medical University, China
2Beijing Retinal and Choroidal Vascular Study Group, China
3Save Sight Institute, University of Sydney, Australia
Abstract
Cholesterol esters, synthesized from cholesterol with long-chain fatty acids, are essential components of plasma lipoproteins and cell membranes that participate in various metabolic processes in the body. Cholesterol can be excreted through the cholesterol reverse transport pathway when excessive cholesterol is produced in the extrahepatic cells, which is regulated by the liver X receptor and its downstream regulators ATP-binding cassette subfamily A member 1 and ATP-binding cassette subfamily G member 1 genes. Abnormal cholesterol metabolism is closely associated with the development of diabetic retinopathy . However, the precise underlying mechanism of the RCT pathway in the pathogenesis of DR is still not fully understood. This review focused on cholesterol metabolism, with a particular emphasis on the RCT pathway and its correlation with the development of DR. Particular attention has been paid to the key regulators of the RCT pathway: LXR, ABCA1, and ABCG1 genes and their potential therapeutic targets in the management of DR.
1. Introduction
2. Cholesterol: Biosynthesis and Regulation
2.1. Synthesis and Transportation
2.2. Conversion and Efflux
2.3. Liver X Receptors and Metabolism of Cholesterol
Read Also: Does Shrimp Give You High Cholesterol
Which Effects Do Various Drugs Have On Reverse Cholesterol Transport
LXR agonists
PPAR agonists
Peroxisome proliferator-activated receptors are transcription factors that like LXRs belong to the nuclear receptor family and modulate expression of genes implicated in several biological processes such as lipid metabolism, glucose metabolism, and inflammation . Three members of the PPAR family have been identified , which have a distinct tissue distribution and modulate different biological responses after activation . Lately, PPAR ligands have attracted interest in view of their potential use for treatment of cardiovascular diseases. Both in humans and experimental animals activation of PPARs has been associated with a raise in plasma HDL cholesterol levels, which in theory might improve RCT.
Dietary supplementation with a PPAR-specific agonist was associated with an elevated level of macrophage-derived tracer excreted into feces of wild-type mice . Compared with PPAR, which has been shown to modify in vivo RCT at the macrophage level , PPAR-mediated effects on the macrophage RCT pathway seem largely confined to the intestine . PPAR activation in mice led to a decreased intestinal expression of Npc1l1 and as a consequence diminished the capacity of the intestine to absorb cholesterol .
Abca1 Abcg1 And Lxr Data
Data are depicted in figures 4 and 5. The CRF group exhibited a significant increase in ABCA1 abundance in the aorta compared with that found in the sham-operated control rats. Similarly, ABCG1 abundance in the thoracic aorta was markedly elevated in the CRF group. Upregulation of ABCA1 and ABCG1 transporters in the aorta of CRF rats was accompanied by significant rise in the abundance of their master regulator, LXR. These events represent a compensatory response to increased cellular lipid burden aimed at enhancing extrusion of surplus lipids by upregulating the efflux pathway.
Fig. 4
Representative Western blots and group data depicting protein abundance of the aorta tissue ABCA1 and ABCG1 in the CRF and CTL groups . * p < 0.05.
Fig. 5
Representative Western blots and group data depicting protein abundance of the aorta tissue LXR/ in the CRF and CTL groups . ** p < 0.01.
Read Also: Shrimp Has High Cholesterol
Clinical Investigations On Rct And Cholesterol Efflux
A variety of evidence shows that RCT and cholesterol efflux play a major role in preventing atherosclerosis in humans. In fact, congenital impairment in genes involved in cholesterol efflux may augment atherogenesis in some patients. On the other hand, acceleration of RCT and cholesterol efflux by increasing HDL or apoA-I levels may result in amelioration of atherosclerosis, suggesting a potential therapeutic tool for human atherosclerosis.
Animal Model And Treatment
C57BL/6J male mice were divided into four groups and, respectively, fed with chow diet or western diet for 12 weeks. Canagliflozin was mixed with chow diet or western diet at 0.03% .22 Fecal samples and urine were collected for three consecutive days using metabolic cages before being killed. Mice were killed through CO2 after fasted overnight. Blood, liver, duodenum, jejunum and ileum were collected and stored in 80°C. All mice were housed in the institutional animal care of West China Hospital of Sichuan University with 24°C-indoor temperature, 55±15% relative humidity, and a 12-hour light/dark cycle. Mice were bred in standard cage with ad libitum feeding. All animal experiments in this research were carried out in accordance with the relevant regulations of the Sichuan University Laboratory Animal Ethics committee and approved by the Institutional Review Board of the Sichuan University Laboratory Animal Ethics committee .
Also Check: Is Shrimp Bad For Your Cholesterol
Cana Augment The Intestinal Abcg5/8 Expression
Intestinal cholesterol absorption and transport also play a vital role in cholesterol homeostasis.25 On the apical, cholesterol is transported into the enterocytes by Npc1l1.5 Cana treatment had little effect on the expression of Npc1l1 . Cholesterol in the enterocytes either transports back into the intestinal lumen by abcg5/8 or transports into basolateral by Abca1 and Abcg1.26 The expression of Abcg5/8 was upregulated 23 times than that in the Cana treated duodenum and jejunum , suggesting higher efflux of cholesterol back to the lumen. The expressions of Abca1 and Abcg1 were also higher 1.52 times in the duodenum and jejunum . Apo A-I receives cholesterol efflux from Abca1, then participates in new HDLs synthesis.27 The expression of Apo A-I was consistently increased in the duodenum and jejunum of Cana treatment western diet fed mice . As a result of elevated efflux of cholesterol, the fecal cholesterol was consistently higher in Cana treated mice . LXR agonist upregulates Abca1, Abag1 and Abcg5/8 expression, and accelerates biliary and fecal cholesterol disposal.24,28 The mRNA expression of LXR was also upregulated in intestine . We also tested the effect of Cana in human colon adenocarcinoma cell line, Caco2 cells. In Caco2 cells, both mRNA and protein expression of Abcg5 and Abcg8 were higher about 1.52 times after Cana treatment . And the LXR expression was also significantly increased by about 2.5 times by Cana .
Which Proteins Impacting Cholesterol Transport Through The Plasma Compartment Are Relevant For Reverse Cholesterol Transport
The association between HDL cholesterol levels and macrophage-specific RCT is less straightforward. Hepatic and intestinal ABCA1 are crucial for HDL particle maturation, and mice with targeted deletion of ABCA1 have almost no circulatory pool of HDL . In agreement with the lack of HDL, ABCA1 knockout mice exhibit an overall defect in macrophage-specific RCT . However, the anti-atherosclerotic compound probucol, that inhibits hepatic ABCA1 activity and thereby reduces HDL cholesterol, had no effect on macrophage RCT in wild-type mice and even increased the flux through the macrophage RCT pathway on the SR-BI knockout background . As one possible explanation the authors hypothesized that treatment with probucol targeted HDL-derived cholesterol taken up into the liver for biliary excretion by preventing hepatic ABCA1-mediated resecretion of cholesterol into the circulation . Another example of a dissociation between plasma HDL cholesterol levels and macrophage RCT are SR-BI knockout mice . Thus, plasma HDL levels do not necessarily represent a reliable reflection of macrophage RCT rates, and for that reason HDL cholesterol levels should be used with caution as a surrogate for predicting fluxes through the RCT pathway.
You May Like: Is Pizza High In Cholesterol
Transintestinal Cholesterol Excretion And Fc Bioavailability
The relative contributions of spontaneous and SR-B1mediated transfer to TICE are unknown. However, TICE is probably only one component of FC transfer to multiple tissue sites. Although our data showed rapid hepatic FC uptake in mice over longer time intervals, the FC appeared in nearly every tissue site, especially erythrocytes .38 Thus, FC is highly mobile and goes to any tissue or cell site by diffusion.53