Pfo Binding Decreased The Cholesterol Accessibility At The Membrane
One explanation for the differential binding observed for pPFO and pPFOD434S in model and natural membranes is that the pPFO derivative binds more cholesterol molecules than pPFOD434S. If this is the case, for the same number of cholesterol molecules one would expect more pPFOD434S molecules associated with the membrane than that of pPFO. Alternatively, if both proteins bind the same number of cholesterol molecules, the differential binding could be explained by the different cholesterol threshold. Cholesterol accessibility at the membrane is progressively reduced by the binding of each protein molecule. Binding of a PFO derivative will continue up to the point where the accessibility of cholesterol drops below its particular binding threshold, for example, below 35mol% cholesterol for the model membrane-PFO combination used in Fig. .
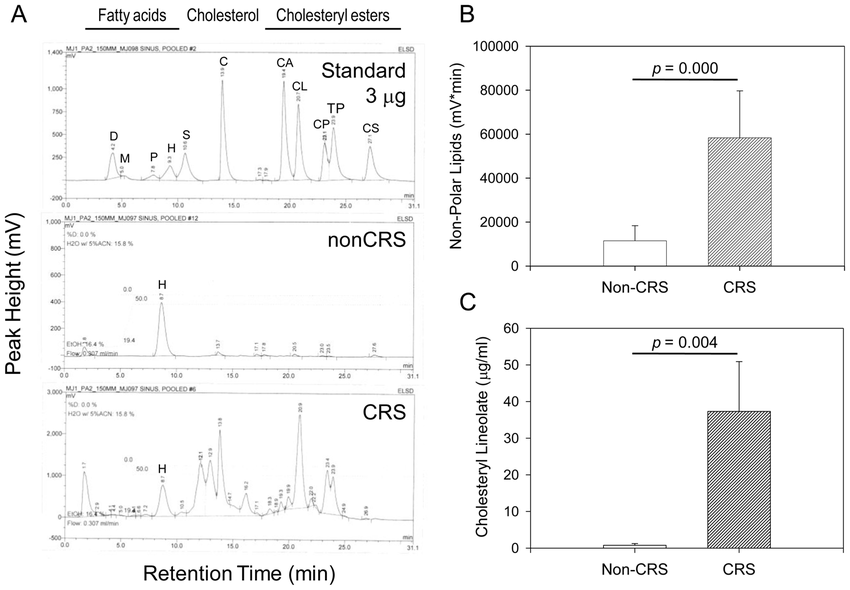
Figure 5
Plasma Transport And Regulation Of Absorption
As an isolated molecule, cholesterol is only minimally soluble in water, or hydrophilic. Because of this, it dissolves in blood at exceedingly small concentrations. To be transported effectively, cholesterol is instead packaged within lipoproteins, complex discoidal particles with exterior amphiphilic proteins and lipids, whose outward-facing surfaces are water-soluble and inward-facing surfaces are lipid-soluble. This allows it to travel through the blood via emulsification. Unbound cholesterol, being amphipathic, is transported in the monolayer surface of the lipoprotein particle along with phospholipids and proteins. Cholesterol esters bound to fatty acid, on the other hand, are transported within the fatty hydrophilic core of the lipoprotein, along with triglyceride.
There are several types of lipoproteins in the blood. In order of increasing density, they are chylomicrons, very-low-density lipoprotein , intermediate-density lipoprotein , low-density lipoprotein , and high-density lipoprotein . Lower protein/lipid ratios make for less dense lipoproteins. Cholesterol within different lipoproteins is identical, although some is carried as its native “free” alcohol form , while others as fatty acyl esters, known also as cholesterol esters, within the particles.
Tuning The Ppfo Cholesterol Threshold By Amino Acid Substitutions In The Membrane Binding Domain
To quantify the effect that amino acid substitutions have on the cholesterol threshold of PFO, we defined the term mol% cholesterol at half binding or just mol% for simplicity. This is the difference in mol% cholesterol between the concentration of cholesterol required for half binding for the PFO derivative under analysis, and the one for nPFO measured using the same membranes. The use of a relative quantity , instead of the actual mol% at half binding, eliminated the differences that may arise from small variations in the lipid composition of different preparations of liposomes. In our hands, the absolute cholesterol mol% at half binding for a PFO derivative could vary approximately up to 3mol% when two independent liposome preparations are compared. These variations are caused by small differences in sample handling when preparing liposomes and lipid solutions. It is therefore important to emphasize that to determine a mol%, the binding isotherms need to be obtained using the same liposome preparation. A PFO derivative that requires more cholesterol for binding than nPFO will show a positive mol% value, while a negative mol% will indicate a derivative that requires less cholesterol for binding. For example, the mol% for rPFO is +4, indicating that for half binding this derivate will required 4mol% more cholesterol in the membrane than nPFO .
Read Also: Cholesterol In Pork
Maturation Stage Of Crbcs
Figure 2. Maturation stage of nRBCs, nRETs, cRBCcb, cRBCpb and cRBCpb+lipids determined by microscopic evaluation after New Methylene Blue staining. Representative pictures of cells from all sources. Bright field, oil, 100 Ã magnification . Dependent on the remaining RNA network, cells are classified into stages 0 to V. Stage 0 shows the typical reticular mesh-like structures of immature cells stained in dark blue, while stage V represents fully mature erythrocytes without any dark blue reticular staining. Diagram showing the affiliation of cells for the different maturation stages in nRBCs, nRETs, cRBCcb,cRBCpb and cRBCpb+lipids displayed in%.
Analysis Of Crystallographic Structures Containing Cholesterol Sulfate
The analysis of the few available crystal structures of cholesterol may not be enough to define common features of cholesterol sites in soluble proteins. Thus, we also studied cholesterol sulfate. This is a cholesterol derivative in which the 3-hydroxyl is substituted by a sulfate group. Although this substitution diminishes the overall hydrophobicity of the molecule, the remaining structural features are identical to those of cholesterol. From a biological standpoint, cholesterol sulfate has been extensively recognized as one of the most important sulfonated steroids. Higher levels of cholesterol sulfate were found in the plasma of patients with liver cirrhosis and hypercholesterolemia while atherosclerosis has been linked to cholesterol sulfate deficiency . Under normal physiology, cholesterol sulfate plays a critical role in platelet adhesion and keratinocyte differentiation. At the molecular level, this steroid regulates the activity of serine proteases and, in a rather selective manner, of protein kinase C isoforms. Several PDB entries describe cholesterol sulfate-protein complexes. We analyzed three complexes found in the PDB database, their topology being depicted in . Averaged volume of the site was estimated at 2,745 ± 256 Å3. The key features of cholesterol sulfate binding sites are summarized in .
Also Check: How Do Cheerios Lower Cholesterol
Lipid Composition Of Crbcs
Quantitative comparison of the lipid content of RBCs was performed by high-resolution mass spectrometry. In nRBCs, cholesterol represented the largest fraction, with 49.2 ± 9.9% of the total lipid content, followed by PC and SM . The sum of all the other lipids constituted the remaining 13.5 ± 3.1% . Interestingly, exactly the same pattern was found in nRETs, although the absolute content of lipids was significantly higher than that in nRBCs , in line with the larger cell size and membrane surface . In more detail, in nRETs higher absolute amounts were observed for each individual lipid type with the exception of LPC, reaching statistical significance for PC and PE .
Figure 3. Lipid content measured by high-resolution mass spectrometry in nRBCs, nRETs, cRBCpb, cRBCcb and cRBC after lipid supplementation Proportional lipid content including PE, PC, PS, PI, SM, LPC and cholesterol of nRBCs, nRETs, cRBCpb, cRBCcb and cRBCpb+lipids displayed as pie charts. The magnitudes of the circles correspond to the absolute lipid content of the cell type. Absolute lipid content of nRBCs, nRETs, cRBCpb and cRBCcb and cRBCpb+lipids regarding the phospholipid groups PC, PE, PS, PI, SM, LPC, and cholesterol. Values are depicted as pmol/107 cells .
An Overview Of Transcription And Translation
This short animation from the Discovery Channel provides a nice overview of the transcription and translation. Transcription is the process by which a gene, segment of DNA that encodes for a specific protein, serves as a template for the synthesis of a messenger RNA for that specific protein. Transcription takes place inside the cell nucleus where chromosomal DNA is located. The mRNA then leaves the nucleus through special pores in the membrane of the nucleus. Once the mRNA emerges from the nucleus, it attaches to a two part structure called a ribosome, which consists of ribosomal RNA . Enzymes also attach to the ribosomal complex and aid in the process of translation, in which the coded sequence of bases on the mRNA is translated and directs the synthesis of a chain of amino acids, which are the building blocks of proteins.
Don’t Miss: Are Mussels High In Cholesterol
Fatty Acids And Triacylglycerides
The fatty acids are lipids that contain long-chain hydrocarbons terminated with a carboxylic acid functional group. Because the long hydrocarbon chain, fatty acids are hydrophobic or nonpolar. Fatty acids with hydrocarbon chains that contain only single bonds are called saturated fatty acids because they have the greatest number of hydrogen atoms possible and are, therefore, saturated with hydrogen. Fatty acids with hydrocarbon chains containing at least one double bond are called unsaturated fatty acids because they have fewer hydrogen atoms. Saturated fatty acids have a straight, flexible carbon backbone, whereas unsaturated fatty acids have kinks in their carbon skeleton because each double bond causes a rigid bend of the carbon skeleton. These differences in saturated versus unsaturated fatty acid structure result in different properties for the corresponding lipids in which the fatty acids are incorporated. For example, lipids containing saturated fatty acids are solids at room temperature, whereas lipids containing unsaturated fatty acids are liquids.
A triacylglycerol, or triglyceride, is formed when three fatty acids are chemically linked to a glycerol molecule . Triglycerides are the primary components of adipose tissue , and are major constituents of sebum . They play an important metabolic role, serving as efficient energy-storage molecules that can provide more than double the caloric content of both carbohydrates and proteins.
A Perspective On Size
An angstrom is one ten-millionth of a millimeter, or 1×1010 meters. The illustration below gives an idea of the relative scale of some of the biological structures discussed above.
The distance between two carbon atoms in a fatty acid chain is a little over one angstrom. A glucose molecule is about 9 angstroms. Bacteria are tens of thousands of angstroms. And as a rough estimate, a typical human cell might be approximately 1/100th of a millimeter which is about 1/10th the width of a human hair. For an intriguing perspective on the size of things from the smallest to the largest objects in the universe, take a look at http://htwins.net/scale2/.
Despite their microscopic size, cells have a lot going on all the time. Diagrams and photomicrographs depict cells as rigid, static sacs that are frozen in time, but if we could somehow take a trip inside a cell, we would be staggered by the beauty, complexity, and incredible activity. You can get at least a glimpse of the inner life of a cell by watching the Harvard University animation, “The Inner Life of a Cell” , showing leukocyte activation in inflammation. Some of the terms used in the video will be foreign to you, but the video provides a magnificent sense of the inner works of cells, and it shows cells to be dynamic structures in which many processes are taking place continually.
Read Also: How Much Cholesterol In A Baked Potato
Phospholipids And Biological Membranes
Figure 2. This illustration shows a phospholipid with two different fatty acids, one saturated and one unsaturated, bonded to the glycerol molecule. The unsaturated fatty acid has a slight kink in its structure due to the double bond.
Triglycerides are classified as simple lipids because they are formed from just two types of compounds: glycerol and fatty acids. In contrast, complex lipids contain at least one additional component, for example, a phosphate group or a carbohydrate moiety . Figure 2 depicts a typical phospholipid composed of two fatty acids linked to glycerol . The two fatty acid carbon chains may be both saturated, both unsaturated, or one of each. Instead of another fatty acid molecule , the third binding position on the glycerol molecule is occupied by a modified phosphate group.
Figure 3. Phospholipids tend to arrange themselves in aqueous solution forming liposomes, micelles, or lipid bilayer sheets.
Some Other Related Questions
What are valence electrons?
Valence electrons are those electrons that are present in the outermost shell of an atom that participates in the formation of a chemical bond.
A bond formation needs to contribute at least one valence electron from both atoms. These are the electrons that make weak hydrogen bonds and shared strong covalent bonds in the polar and non-polar molecules.
The structural difference between saturated and unsaturated fatty acids.
The basic structural difference between saturated and unsaturated fatty acids is that the saturated ones are straight chains.
Whereas, the unsaturated fatty acids are bent because of the absence of two hydrogen atoms and the existence of a double bond between carbon atoms. The unsaturated fatty acids are more stable than the saturated ones.
Also Check: Is Pizza High In Cholesterol
Amino Acid Substitutions In Domain 3 Modify The Cholesterol
As described above, modifications to the PFO D4 alter the cholesterol threshold of the protein. When the F318A change was introduced in the distal D3 of the protein to generate the FPFO derivative , no variations in the cholesterol threshold were observed. The substitution of a second aromatic residue in D3 unexpectedly reduced the cholesterol threshold of the protein to levels like those observed for the wild type nPFO . Given the long distance that separates Y181 from the D4 tip, it is clear that modifications outside the conserved loops can alter the cholesterol binding properties of PFO derivatives, presumably through the coupling between D3 and D4,. We have therefore generated a parental non-lytic PFO derivative , with the same cholesterol threshold as the wild type protein .
Figure 3
Lipids: Structures And Properties

Defining lipids.Classification of lipids. Properties of lipids and relevance to biological membranes, and biosignaling.
Lipids are a class of molecules – largely derived from biological sources – that have a significant nonpolar character. Thus, one criterion for a lipid is solubility in nonpolar solvents. However, most lipids are not completely nonpolar, but include at least a small polar or charged component or functional group. This gives them a more or less dual nature – both nonpolar and polar – a characteristic termed amphipathic.
Fats, fatty acids, soaps.
Fatty acids are carboxylic acids, with the functional group at one end of a rather long, unbranched hydrocarbon chain. Fatty acids are thus amphipathic – the carboxylic acid is ionized at physiological pH and interacts well with a polar solvent , while the hydrocarbon chain is quite nonpolar, and contributes a hydrophobic effect.
Triacylglycerols
Triacylglycerols are a common, simple type of lipid consistingof three long-chain fatty acidsesterified to glycerol, a three-carbon triol. They are the typical storage form of fats in adipose tissue, providing about 9 kcal/g of energy.
The figure at right shows a typical triacylglycerol as a structural formula. Carbon atoms are indicated at vertices and ends of lines. Hydrogen atoms are implicit. The three-carbon glycerol backbone is drawn in black. Three fatty acids are shown in ester linkages with glycerol.
Phospholipids and membranes
Cholesterol
Related topics pages:
Read Also: Are Pork Chops Heart-healthy
Degradation Of Fatty Acids With An Odd Number Of Carbon Atoms
- Final cycle of fatty acid oxidation
- Degradation of fatty acids with an even number of carbon atoms yields two acetyl-CoA .
- Degradation of fatty acids with an odd number of carbon atoms yields one acetyl-CoA and one propionyl-CoA .
Ketone body synthesis takes place exclusively in the mitochondria of hepatocytes. Ketone bodies are then released into the blood and transported to their target tissues .
Two molecules of acetyl-CoA acetoacetyl-CoA HMG-CoA acetoacetate -hydroxybutyrate. Acetone is formed by spontaneous of acetoacetate. The body has no use for acetone, which is excreted primarily via the lungs . A small fraction is also exerted in the urine.
Are All Lipids Non
Yes, all lipids are non-polar in nature all of them make long chains of hydrocarbons through covalent bonding. There are seven major categories of lipids mentioned down below:
- Saturated fatty acids
- Phospholipids
- Steroids
Try googling out any one of them, and you will find the presence of long -CH chains made from an equal number of shared pairs of electrons.
You May Like: Is Deer Meat Fattening
Very High Cholesterol Content
The highest amount of Chol that can dissolve in a PL bilayer is 50 mol% . At a higher Chol/PL molar ratio, Chol is distributed between the Chol-saturated fraction of the PL/Chol bilayer and the CBDs immersed in it. Among biological membranes, such a high Chol content is observed only in the fiber cell plasma membranes of the eye lens, and this is their most unique biochemical characteristic. In the membrane of the human eye fiber cell, the Chol/PL molar ratio ranges from 12 in the lens cortex and can be as high as 3 to 4 in the lens nucleus .
Based on the results of our EPR spin-labeling experiments with lens-lipid membranes as well as those found in the literature, we have been able to identify the functions of Chol specific to the fiber cell plasma membrane . The CBD plays a crucial role in these membranes as it ensures that the surrounding PL bilayer is saturated with Chol. CBDs start to form at 50 mol% Chol an increase of the Chol content above 50 mol% does not change the properties of the surrounding PL bilayer .6C). This feature is independent of the bilayer PL composition as is clearly illustrated in , which shows profiles of the four measured properties across bilayers consisting of lipids obtained from human lens membranes from different donors. For all investigated age groups, appropriate profiles are identical.
What Lipids Or Fatty Acids Are Made Of
The lipids are mostly made of carbon and hydrogen atoms which are bonded with strong sharing covalent bonds.
Because, an equal number proportion of electrons are shared between the carbon and hydrogen atoms, so, there is no partial charge that develops anywhere throughout.
Due to this, non-polar molecules can form long-chain carbon-hydrogen bonds.
Furthermore, each lipid molecule has the presence of a carboxyl group attached to the carbons and also hydrogen atoms attached to this carbon chain.
This carboxyl group is responsible for hydrophobic and hydrophilic behavior during the grease or oil cleaning by soap.
You can also read out a valuable article related to the non-polar nature of oil.
Read Also: Are Baked Potatoes High In Cholesterol